Research identifies a protein that helps determine the fate of RNA
After it is transcribed from DNA, RNA can go on to many fates. While the most familiar path may lead directly to the production of protein, RNA molecules themselves can also become capable of altering the expression of genes. New research helps explain how the destiny of an RNA sequence is achieved.
In a study published August 27 in Cell, Rockefeller University scientists and a colleague at Columbia University have identified a protein that recognizes a chemical instruction tag affixed to an RNA sequence, an important step in the decision-making process.

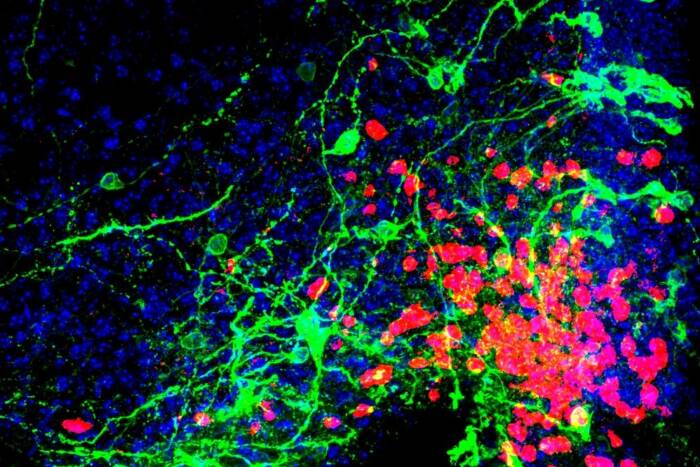
Ready to read: The newly identified protein, HNRNPA2B1 (green), which recognizes the m6A tag, is found within the nuclei (blue) of cells. Actin filaments, important elements of the cell’s structure, show up in red. Credit: Lisa Noble and Gloria Wu
“This tag, known as m6A, was identified more than four decades ago on RNA sequences. Since then, this abundant label has been implicated in several important processes that influence the production of proteins, as well as so-called microRNAs,” says study author Sohail Tavazoie, Leon Hess Associate Professor, and head of the Elizabeth and Vincent Meyer Laboratory of Systems Cancer Biology. (Micro-RNAs are small RNA molecules that do not code for proteins, but instead turn down the activity of genes.)
“However,” Tavazoie says, “a lingering question remained: What reads this chemical tag within the nucleus of cells? Claudio Alarcón, a research associate in my lab, has identified one such “reader” protein. Because of the fundamental nature of the processes involved, this discovery has implications for cells’ normal function and for disease.”
The tag, called m6A, is a methyl group attached to a particular part of an adenosine, a component of RNA’s sequence. In previous work, Alarcón and colleagues identified the m6A tag as an important regulator of the production of microRNAs. The “writer” protein that places this tag was already known to mark RNA molecules that need to be spliced before they are translated into proteins. Many genes contain sections that must be cut out, and the RNA splicing process is crucial to the function and identity of a cell.
The recent experiments show that the newly discovered reader, a protein known as HNRNPA2B1, recognizes m6A tags on RNA destined for two separate fates: Trimming to become microRNAs or splicing for proper production into protein. After recognizing m6A tags on microRNA precursors, HNRNPA2B1 then recruits the cutting machinery responsible for further trimming and processing those RNA molecules. Future work is required to understand how HNRNPA2B1 interacts with the proteins involved in the splicing of RNA.
The researchers suspected that the HNRNPA2B1 protein acts as a reader because they found it frequently binds to the same sites on the RNA molecule where the m6A tag attaches. To determine what the alleged reader was doing there, the team reduced its presence in cells.
In cells with reduced HNRNPA2B1 levels, they found a shift in the expression of microRNAs overall, with many microRNAs reduced. They also looked at the effects on RNA destined for splicing. Here too, they found telling changes in the splicing of different RNA molecules that are dependent on m6A tags.
HNRNPA2B1 is the first m6A nuclear reader to be identified, and evidence from the experiments suggests the existence of additional readers within the nucleus that also recognize this tag.
“The discovery of this new m6A reader has ramifications for a broad range of processes,” Alarcón says. “RNA splicing establishes the repertoire of proteins available in cells. Meanwhile, abnormalities in microRNAs have been associated with several diseases, including cancer. This work also contributes to growing evidence that information beyond the sequence of RNA, such as chemical modifications, can determine the ultimate fate and function of RNA molecules.”
 |
Cell, online August 27, 2015 HNRNPA2B1 Is a Mediator of m6A-Dependent Nuclear RNA Processing Events Claudio R. Alarcón, Hani Goodarzi, Hyeseung Lee, Xuhang Liu, Saeed Tavazoie, and Sohail F. Tavazoie |